|
I am interested in platelet activation in the context of their physiological functions, cardiovascular disorders, and their interactions with biomaterials used in vascular and dental implants. I am also developing cell membrane models with a focus on the aminophospholipid (phosphatidyl serine) asymmetry for basic cell membrane studies as well as the studies of blood coagulation.
This research falls under the broad umbrella concept of biointerfaces, an interdisciplinary field integrating biology, surface physical and analytical chemistry, engineering, and physics. See my recent editorial in Biointerphases on how the face of the biointerfaces field has been changing in recent years.
It relies heavily on a range of biological, biophysical, and surface analytical techniques (ultra-high vacuum as well as "wet"), such as flow cytometry and (immuno/confocal)fluorescence microscopy, X-ray photoelectron spectroscopy (XPS) and Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS), quartz crystal microbalance (QCM) and atomic force microscopy (AFM), neutron reflectometry at solid/liquid and air/liquid interfaces.
|
Blood-biomaterial interactions and biocompatibility: |
|
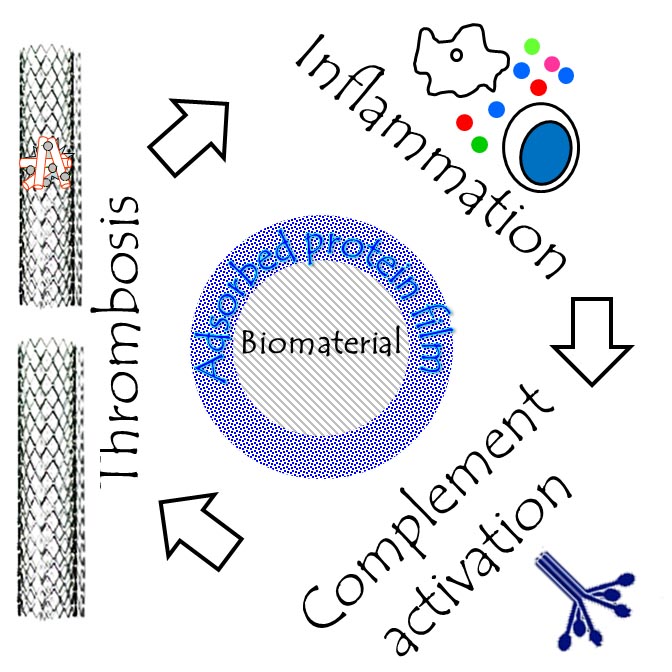
Despite extensive research, all materials used in medical applications today cause thrombotic and/or inflammatory reactions. They are used in combination with anti-thrombotic and/or anti antiplatelet therapy at great cost to the healthcare system and danger to the patient.
We are analyzing coagulatory and immune responses of blood exposed to materials under static and dynamic conditions, protein conformation and protein film composition on the surfaces of clinically relevant materials, and studying molecular mechanisms of platelet activation at biomaterial surfaces.
The image on the left is adapted from Gorbet and Sefton, Biomaterials 25, 5681 (2004).
|
Platelets: |
 |
Key effectors in hemostasis, inflammation and
wound
healing, platelets are also involved in cancer metastasis and adaptive
immune response. How do they manage to perform such diverse functions?
I am interested in the
mechanisms underlying platelet response to different
stimuli and novel
methods
to study these mechanisms. |
Cell
membranes and Solid Supported Lipid Bilayers: |
 |
Membranes of cells are based on lipid bilayers. We use solid-supported lipid bilayers to model cell membranes as well as to study the interactions between model biological systems (lipids) and surfaces of artificial materlas, such as silica, titania, or mica. |
Acoustic Waves in the Limelight: |
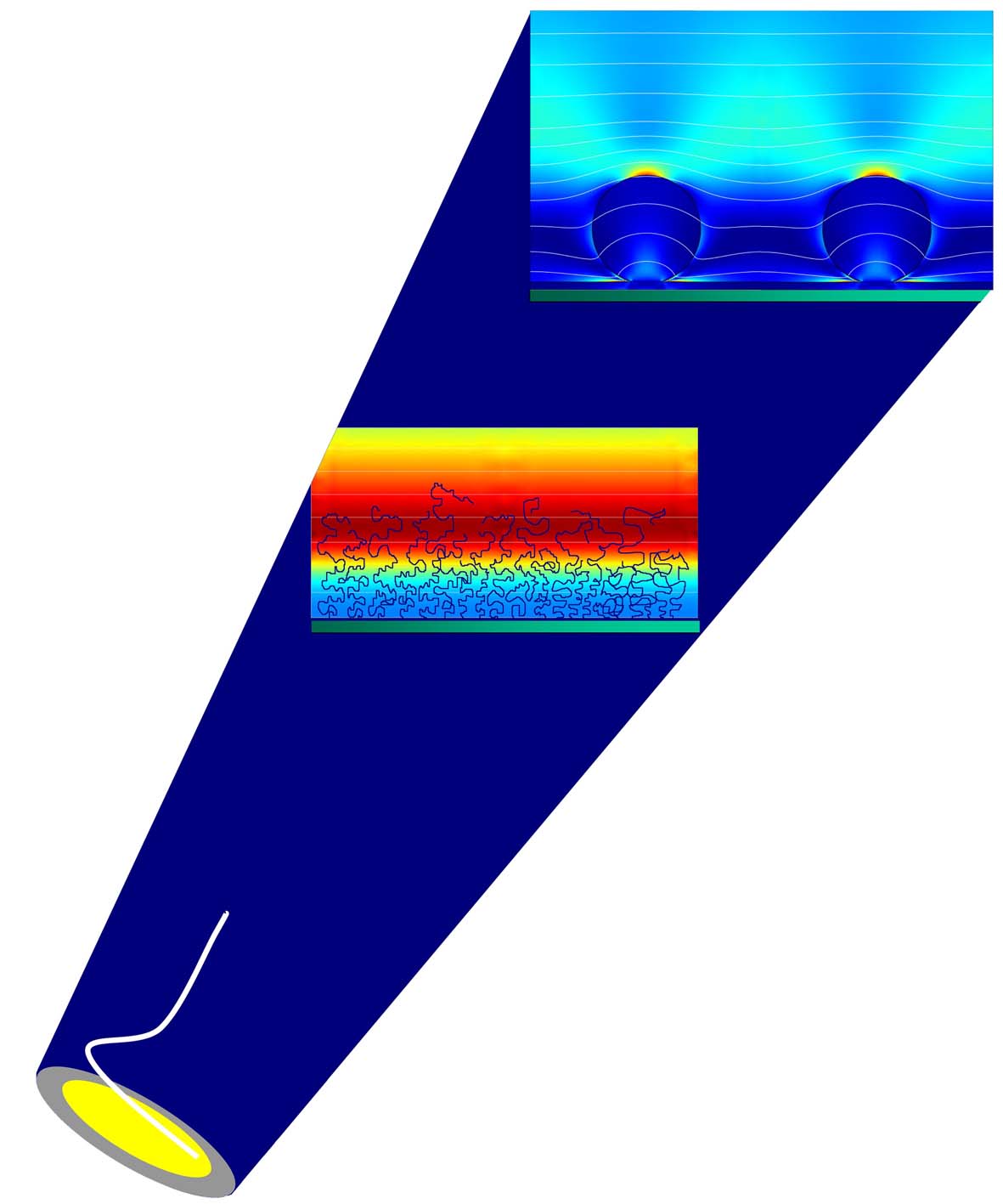 |
Acoustic sensing with quartz crystal microbalance
(QCM) in liquids:
how do acoustic waves interact with thin biomolecular films?
What can
we learn about soft/biological, hydrated interfaces from QCM? |
Protein
crystallization in two and three
dimension (2D and 3D): |
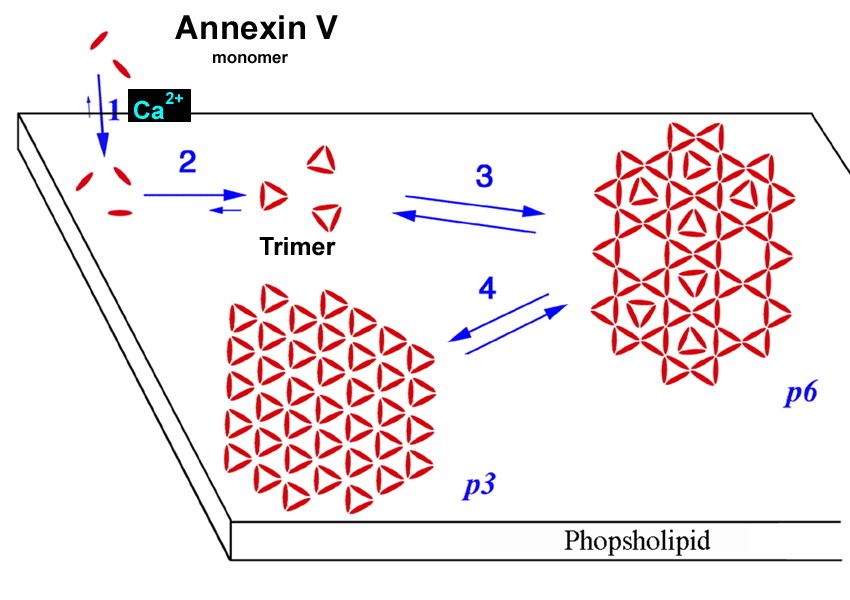 |
Two-dimensional crystals of Annexin A5.
Three-dimensional crystals of insulin.
Studied with high-resolution atomic force microscopy (AFM). |
Titania: |
|
|
Because titania is an important biomaterial, we often use it in our
experiments to study blood, platelets, and lipids at its surfaces. |
Techniques: |
|
 |
Many techniques are used in my laboratory, but I
have particularly
strong background with atomic force microscopy of soft specimens in
liquid (AFM) and quartz crystal
microbalance (QCM), as well as neutron reflectometry (lipids),
secondary ion mass
spectrometry (SIMS) and flow cytometry. |
|
|
Keywords: |
|
|
- Biological Surfaces and Interfaces. Biosurfaces. Biointerfaces. Analysis of soft and hydrated surfaces.
Interactions between biological and artificial systems/materials.
- Blood-biomaterial interactions. Platelet-biomaterial interactions.
Platelet activaton. Platelets in wound healing.
- Titania, metallic implants. Lipid interactions with titania.
- Cell membranes, cell membrane biophysics, cell membrane models,
aminophospholipid asymmetry. Lipid interactions with oxide
surfaces.
- Atomic force microscopy of soft specimens in liquid. Quartz crystal microbalance (QCM). Neutron
reflectometry (lipids). Secondary ion mass spectrometry (SIMS).
- Annexin A5, phosphatidyl serine, insulin, ferritin, apoferritin, Clotting, clotting factors, FVIII.
|
|
|
|
|
|
|
|